BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://journal.zums.ac.ir/article-1-6123-en.html
2- Dept. of Sport Physiology, Faculty of Sports Sciences, Ferdowsi University of Mashhad, Mashhad, Iran ,
3- Parsian Diabetes Clinic, Mashhad, Iran
✅ The analysis showed that only LTRT among the types of exercise can elevate irisin concentration. Therefore, LTRT seems to have more beneficial influences, compared to other modes of exercise in inclusive irisin responses.
Although the beneficial effects of exercise are well documented, the molecular mechanisms by which exercise prevents chronic diseases and improves health are poorly understood. Skeletal muscles have been identified as an active endocrine organ. They have critical functions in metabolic homeostasis by releasing hormones, called myokine, during muscle contraction (1). Myokines mediate crosstalk between tissues, such as the adipose tissue, liver, skin, and pancreas. They locally affect muscles and impose metabolic impacts through autocrine, paracrine, and endocrine signaling (1).
Irisin seems to be predominantly involved in exercise adaptations as a contraction-regulated myokine. It has sufficient potential to prevent and treat obesity and metabolic diseases (2, 3). Irisin regulates energy metabolism and thermogenesis, increments white adipocytes browning (fat browning), improves insulin sensitivity, and upgrades body compositions (4). Moreover, irisin promotes mitochondrial biogenesis and effectively increases oxidative capacity (2, 3). It is produced following proteolytic cleavage and the glycosylation and dimerization of fibronectin type III domain-containing protein 5 (FNDC5), which is set via peroxisome proliferator-activated receptor (PPAR) gamma coactivator 1α (PGC-1α) (5, 6).
It has been shown that acute or long-term exercise can induce PGC-1α expression in tissues resulting in the induction of FNDC5 production and increases irisin secretion as a myokine (7). Although various studies introduce exercise as an irisin-augmenting factor in rats (2, 8, 9), conflicting results have been reported about humans (10, 11). However, Bostrom et al. (2012) revealed irisin elevation following exercise in humans (12). In this regard, some investigations reported that short-term moderate-intensity continuous training (MICT) or high-intensity interval training (HIIT) leads to a transient increase in irisin concentration (13-15). Moreover, irisin raise has been observed in various studies in response to long-term aerobic training (AT) or resistance training (RT), while no significant impact has been reported on irisin as the result of exercise (11, 16, 17). Furthermore, it is well known that muscle contraction is essential to prevent metabolic diseases. However, the hormonal and physiological adaptations of AT and RT are different (18-20). In addition, several systematic reviews and meta-analyses have examined irisin concentration regulation by exercise. None of the previous papers included critical analyses of different modalities of exercise. In other words, it is not clear which kind of exercise is more effective in irisin concentration. Finally, this paper aimed to perform a meta-analysis to define the possible influential types of exercise training on irisin concentration in adults.
Data Sources and Searches
All English databases, including PubMed, CINAHL, Google Scholar, Medline, and Scopus were searched. The following phrases and words were searched in the keywords, titles, abstracts, introduction, subjects, and heading of articles: irisin, FNDC5, PGC1-α, exercise, physical activity, training, aerobic training, RT, concurrent training, short-term exercise training, and long-term exercise training. The Boolen search strategy was used (AND, OR, or NOT). All articles including meta-analyses, systematic reviews, and the references of the used articles were searched and assessed until April 25, 2020.
Study Selection
All types of exercise, including long-term aerobic training (LTAT), long-term resistance training (LTRT), and short-term resistance training were selected randomly in the pre- and post-test conditions. In our meta-analysis resistance exercise group entailed full-body training with a machine and weight-bearing encompassed at least six movements. Moreover, the aerobic exercise group included walking, running, and aerobic training. One of the authors evaluated the titles, treatment guidelines, summaries, and full-texts of appropriate studies.
Inclusion/Exclusion Criteria
The studies with the following criteria were selected: (1) full-text experimental and controlled trials published in English, (2) participants aged ≥18 years, (3) irisin was one of the main subjects of the study and was measured by enzyme-linked immunosorbent assay (ELISA), (4) the protocol of studies used AT, RT, or combined AT and RT (with an intervention period ≥8 weeks for long-term studies), and (5) inactive to moderately active subjects.
Studies were excluded if they met one of the following conditions: (1) review article or literature review, (2) studies with non-human participants, (3) conferences, (4) abstracts, (5) study protocols in which the participants took part in a training plan within the last six months.
Outcome Measures
The studies that had measured dynamic irisin by different ELISA kits were eligible for the present review.
Data Extraction
Titles and abstracts were assessed separately by an author (MMR). All the authors (MMR, ND, SRAH, and ARS) extracted three types of variable, including participant (study population, sample size, age, and baseline body mass index), exercise protocol details (duration, modalities, intensities, and frequency of exercise), and outcomes (irisin concentrations associated with exercise modalities). The methodology qualification of each study was evaluated separately by an author (MMR) through the Cochrane collaboration ‘risk of bias’ tool (21, 22).
Data Synthesis
According to all investigations, the effect size was reported for outcomes by calculating the mean differences between pre- and post-test. All outcomes were assessed independently. To compute the mean difference of irisin level, some parameters, including sample size, characteristics of the participants, methods of blood analysis, and information of exercise interventions were evaluated by the same methods in pre- and post-test conditions.
All data were assessed by Review Manager 5.3 (The Nordic Cochrane Center, Copenhagen, Denmark). The post-test mean was subtracted from the pre-test mean and the standard error of means (SEM) was changed to standard deviation. TA Tech Tip and GetData Graph Digitizer software were used for any data which had not been shown in the text or tables (displayed data in figures) and we were not able to reach the authors. Therefore, Heterogeneity was calculated as Cochrane’s Q and I2 index >50%. We presented a 5% level of significance for the forest plot to describe the results. A negative effect size demonstrated irisin concentration reduction, while a positive effect size indicated an increase. P-value˂0.05 was considered statistically significant.
Study Quality
The studies were qualified by a fifteen-point tool in exercise reporting (TESTEX) scales (23). Three reviewers (MMR, ND, and SRAH) qualified the studies and reported the assessments.
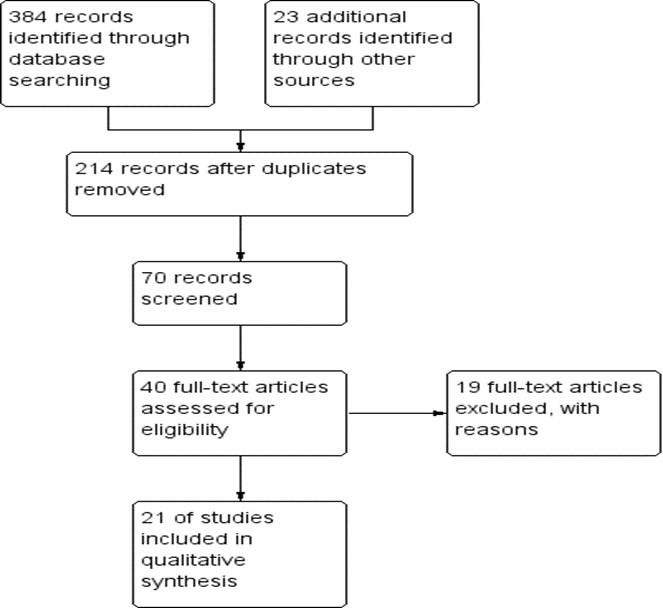
Overall, 384 related articles were found from Scopus, PubMed, Google Scholar, and Science Direct databases. Animal studies and pharmacologic interventions were removed leading to the final number of 214 full-text articles. Seventy studies were evaluated and 21 articles were selected to be reviewed for the moderator variables and stated inclusion criteria (PRISMA flow diagram, Figure 1). These 21 studies included 631 individuals in pre- and post-test. Moreover, 457, 298, 159, and 174 participants were in the aerobic training (short-term + long-term aerobic training) (SLTAT), short-term aerobic training (STAT), LTAT, and LTRT groups, respectively.
Figure 1. The PRISMA flow chart
Intervention Details
Studies duration range was 24 hours to 12 weeks. Full article characteristics have been shown in Table 1. The aerobic exercise training interventions had a range of 30-60 min with an intensity of 50%-85% VO2max. In the resistance exercise intervention, it ranged from 40%-90% 1RM.
Table 1. The characteristics of the investigated articles
| 3 | irisin | irisin | irisin | iriin | irisin | irisin | irisin | irisin |
| Intervention group: frequency and duration |
Maximal workload 70%vo2max /10 min | two AT (3 days among tests) to 64-76% of HR | 30 min. warming, 10 min. stretching and 80 min. handball training | at baseline 45 min cycling (70% VO2max) and combined AT and RT after 12 weeks | 4 weeks HIIT, MICT | MICT at 55% of VO2max, and HIIT 4 min (at 80% of VO2max)/3 min (at 50% of VO2max) | 60minutes by treadmill (20 min warm-up LIT, 6×5 interval training) | 30-minute aerobic exercise |
| Modes of exercise | Short-term aerobic | short-term aerobic | long-term | long-term aerobic | long-term aerobic | short-term aerobic | Short-term aerobic | Short-term aerobic |
| N EX |
35 | 30 | 19 | 26 | 14 | 11 | 9 | 122 |
| gender | F 15 M 20 | M | M | M | 4 F 10 M | F | 2 F 7M | F 61 M 61 |
| Disease | None | None | None | None Overweight | T2DM | obese | None | None |
| BMI (mean ± SD) |
22.4±2.5 | 21.3±0.4 | 23.089±1.090 | NW 23.5±2.0 OW 29.0±2.4 | 27.8 ± 2.8 | 37.3±2.1 | 24.5±2.4 | NM |
| Age (mean ± SD) |
23.0±3.3 | 19.2±0.7 | 16.052±0.848 | 40-65 | 59.6 ± 5.7 | 24.3±1.4 | 32±9 | 20.0 0.1 |
| study | Daskalopoulou et al. (24) | Algul et al.(25) | Dundar et aql. (26) | Norheim et al. (27) | Dunnwald et al. (28) | Winn et al. (29) | Nygard et al. (15) | Anastasilakis et al. (30) |
| Assessment measure/units | irisin | irisin | irisin | iriin | irisin | irisin | irisin | irisin |
| Intervention group: frequency and duration |
Bruce’s protocol was conducted from Monday to Friday during 2 weeks | an acute exercise test at an intensity corresponding to 80% of their VO2max for 50 min. | 90 min treadmill training with 54 min at 60 % of VO 2 max | Cycling at 65% of HR for 40 min. 1 min training and 1 min rest classified by 6 rounds at 85–95% HRpeak. | 10 session in cryogenic chamber | RT protocol included lower muscles training and core muscles | 26 exercises were divided into 5 sets with 10-15 movements at 60-80% 1RM for 8 weeks. | LRT (40%-60%1RM and 20-30 repetition) HRT (70%-90% 1RM and 5-15 repetition) |
| Modes of exercise | Short-term aerobic | short-term aerobic | Short-term aerobic | Long-term aerobic | Short-term aerobic | Long-term resistance | Long-term resistance | Long-term resistance |
| N EX |
38 | 17 | 12 | 30 | 12 | 10 | 7 | 21 |
| gender | F | F 9 M 8 | F 5 M 7 | F | M | M | M | F |
| Disease | None | None |
None | Obese | Obese | None | None | None |
| BMI (mean ± SD) |
22 ± 5.6 | 22.0±1.1 | F 5.2 ± 4.97 M 4.29±2.94 | HIIT 31.65±4.83 A 32.67±5.91 | HFL 30.1±4.0 LFL 35.7±4.5 | 26.18±5.5 | 27.13±2.16 | LRT 23.8±1.58 HRT 23.6±1.57 |
| Age (mean ± SD) |
23 ± 3.3 | 27.4±3.8 | F 3.8 ± 4.7 M 2.71±1.6 | HIIT 16.13±1.64 A 15.47±1.73 | 38.4±8.2 | 62.3 ± 3.5 | 29.37±5.14 | LRT 25.27±3.34 HRT 23.50 ± 2.27 |
| study | Saldaña et al. (31) | Qiu et al. (13) | Kraemer et al. (32) | Herrera et al. (33) | Dulian et al. (34) | Zhao et al. (35) | Bang et al. (16) | Moienneia et al. (36) |
| Assessment measure/units | irisin | irisin | irisin | iriin | irisin | |||
| Intervention group: frequency and duration |
72sessions, lower limbs training with 60%-70%1RM | 16 weeks, 10 exercises, 3 sets, 6-12 repetitions maximum, 30-s rest between sets | 4weeks, 3 consecutive 30-s maximal pedaling exercise with 10-min rest between sets | 8 weeks, 60-min 6 movements, 65-80 % 1RM, 10-12 repetitions for 3 sets (2-3 s concentric, 2-3 s eccentric) | 12 weeks(warm-up, strength elasticity training for 40 min cooldown) | |||
| Modes of exercise | Long-term resistance | Long-term resistance | Long-term resistance | Long-term resistance | long-term resistance | |||
| N EX |
18 | 49 | 20 | 10 | 18 | |||
| gender | 11female 7 male | female | male | 3female 7male | female | |||
| Disease | hemodialysis | NM OW | None | OW | None | |||
| BMI (mean ± SD) |
F 24.2 ± 2.7 M23.5 ± 3.9 | NW 24.3±3.6 OW 30.9±3.1 | 21.0±2.9 | 27.0±3.4 | 25.28 | |||
| Age (mean ± SD) |
F50.9 ± 6.6 M44.8 ± 14.1 | NW 68.0 ± 6.2 OW 66.5 ± 5.0 | 20.9±1.3 | 26.4±2.9 | 74.45±0.62 | |||
| study | Moraes et al. (37) | Tibana et al. (11) | Tsuchiya et al. (38) | Kim et al. (10) | Kim et al. (39) |
Outcomes Measures
Change in Irisin
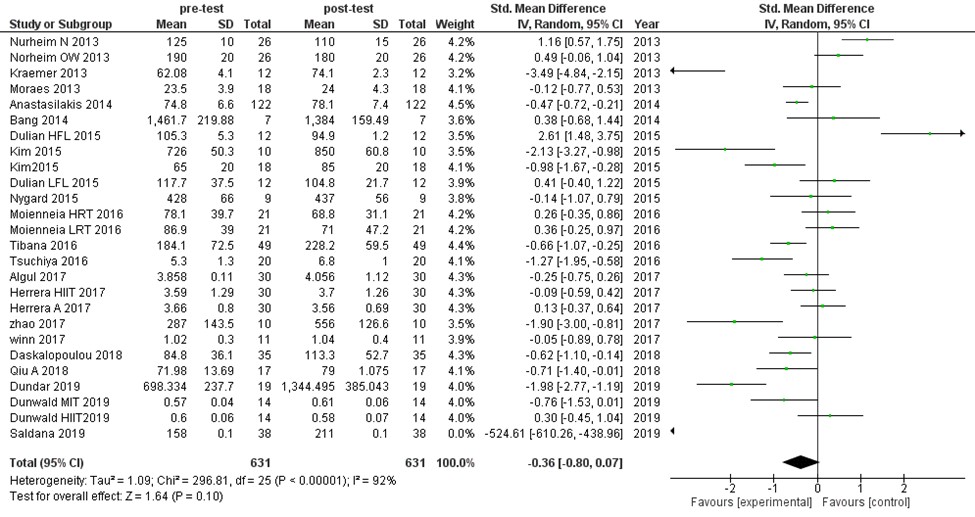
Irisin changes have been reported by 21 studies, which included a total of 631 members (26 pre- and post-tests). We mixed the outcomes to make the random-effects model. No considerable difference in irisin level was observed in the post-test training (MD:-0.36 ng/mL, 95%CI [-0.80, 0.07], P=0.1) (Figure 2).
Figure 2. The forest plot for irisin level in exercise intervention
Exercise Modality Investigation
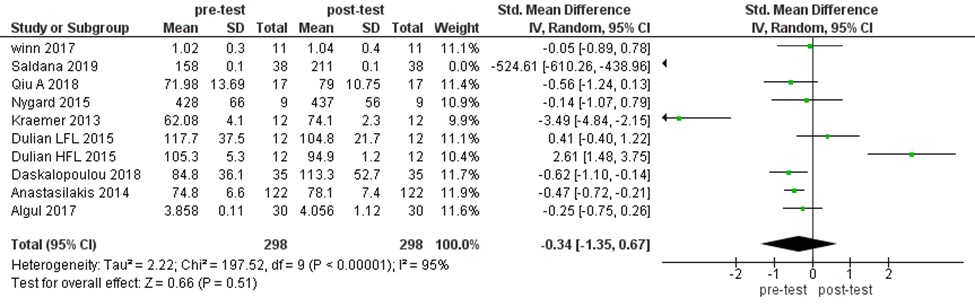
We assessed the effects of intervention training period (i.e., LTAT, STAT, and LTRT) on irisin level. This analysis found that irisin level had not been changed by SLTAT significantly (MD:-0.21 ng/ml, 955 CI [-0.83, 0.4], P=0.49). The obtained values were as LTAT (MD:-0.08 ng/mL, 95% CI [-0.72, 0.57], P=0.82) and STAT (MD:-0.34 ng/mL, 95% CI [-1.35, 0.67], P=0.51). However, the values may alter in LTRT significantly (MD:-0.61 ng/mL, 95% CI [-1.12, 0.09], P=0.02). In contrast, the irisin level increased after LTRT (Figures 3- 6).
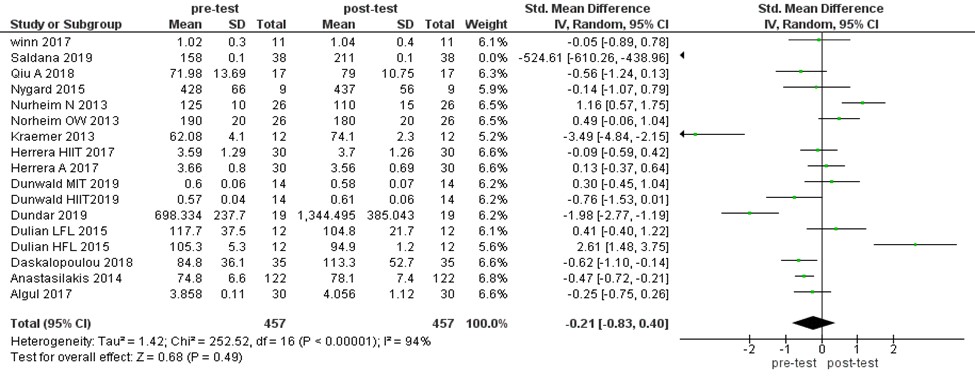
Figure 3. The forest plot of irisin level in an aerobic training group
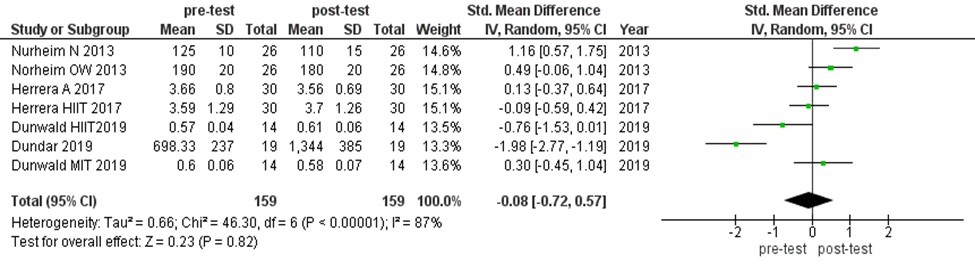
Figure 4. The forest plot of irisin level in LTAT group
Figure 5. The forest plot of irisin level in STAT group
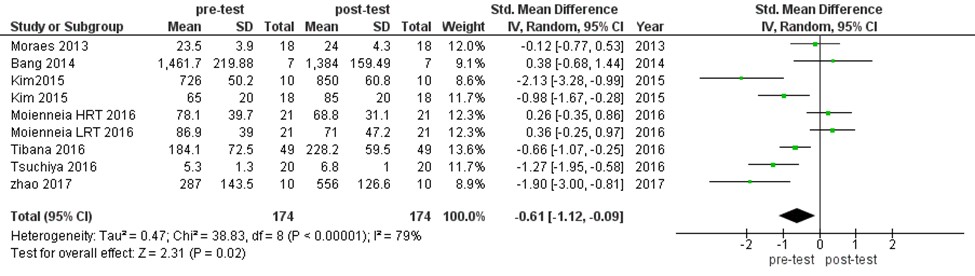
Figure 6. The forest plot of irisin level in LTRT intervention
Table 2. subgroup searching result in meta-analysis
| Variables | Type of exercise | All | |||
| Aerobic | Long-term A | Short-term A | Long-term R | ||
| Irisin | |||||
| N. studies | 13 | 4 | 9 | 8 | 21 |
| Mean difference(MD) | -0.21 | -0.08 | -0.34 | -0.61 | -0.36 |
| 95% CI | [-0.83, 0.40] | [-0.72, 0.57] | [-1.35, 0.67] | [-1.12, 0.09] | [-0.80, 0.07] |
| p-value | 0.49 | 0.82 | 0.51 | 0.02 | 0.10 |
| I2 (%) | 94 | 87 | 95 | 79 | 92 |
Heterogeneity and Publication Bias
The data in all groups revealed high heterogeneity (I2=92%, P=0.1), and the heterogeneity of groups are shown below. The publication risk of bias was determined by Egger plot and it was low (Figure 4).
Study Quality
The quality of all investigated articles was reported by the TESTEX with the results of moderate to good with a median score of 10.14 (range: 7-13) out of the maximum score of 15 (Table 3). All 7, 13, 7, and 5 studies met the criteria of physical activity monitoring, intention to treat analyses, similarities training intensity, assessor blinding criterion, and allocation concealment. In addition, half of the trials met other TESTEX criteria.
Figure 7. Funnel plot of aerobic and resistance exercises
Table 3. This table has been developed to evaluate the quality of articles in the field of exercise studies (TESTEX)
|
Overall TESTEX (15) |
12 |
10 |
10 |
7 |
11 |
10 |
12 |
8 |
12 |
10 |
10 |
7 |
9 |
10 |
12 |
10 |
10 |
10 |
8 |
13 |
12 |
|
Exercise volume & Energy expenditure |
0 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
0 |
0 |
1 |
1 |
1 |
0 |
0 |
1 |
0 |
1 |
1 |
|
Relative exercise intensity constant |
1 |
1 |
1 |
1 |
1 |
1 |
0 |
0 |
1 |
0 |
0 |
1 |
1 |
1 |
1 |
0 |
0 |
0 |
1 |
0 |
1 |
|
Activity monitoring in control group |
0 |
0 |
0 |
0 |
0 |
0 |
1 |
1 |
0 |
0 |
0 |
0 |
0 |
1 |
0 |
1 |
0 |
1 |
1 |
1 |
0 |
|
Point measures & measures of variability |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
|
Reporting between group statistical comparison |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
2 |
|
Intention to treat analysis |
1 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
1 |
1 |
0 |
0 |
0 |
1 |
1 |
|
Outcomes measures assessed > 85% participants# |
3 |
2 |
2 |
1 |
3 |
2 |
2 |
1 |
3 |
2 |
3 |
2 |
1 |
2 |
2 |
2 |
2 |
3 |
1 |
2 |
2 |
|
Assessors blinded |
1 |
0 |
0 |
0 |
0 |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
0 |
0 |
1 |
1 |
0 |
0 |
1 |
1 |
|
Groups similar at baseline |
1 |
1 |
1 |
0 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
0 |
1 |
1 |
1 |
0 |
1 |
1 |
1 |
1 |
1 |
|
Allocation concealed |
0 |
0 |
0 |
0 |
0 |
0 |
1 |
0 |
0 |
1 |
0 |
0 |
0 |
0 |
1 |
0 |
1 |
0 |
0 |
1 |
0 |
|
Randomization details specified |
1 |
1 |
1 |
0 |
1 |
1 |
1 |
0 |
0 |
1 |
1 |
0 |
1 |
0 |
1 |
1 |
1 |
0 |
0 |
1 |
1 |
|
Eligibility Criteria specified |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
|
Study |
Daskalopoulou et al. |
Algul et al. |
Dundar et al. |
Norheim et al. |
Dunwald et al. |
Winn et al. |
Nygard et al. |
Anastasilakis et al. |
Saldana et al. |
Qiu et al. |
Kraemer et al. |
Herrera et al. |
Dulian et al. |
Zhao et al. |
Bang et al. |
Moienneiaa et al. |
Moaress et al. |
Tibana et al. |
Tsuchiya et al. |
Kim et al. |
Kim et al. |
Discussion
This study aimed to investigate the influences of several exercise modalities on irisin concentration in adults. The results showed that LSTAT, LTAT, and STAT did not have any impact on irisin, while LTRT increased irisin concentration in adults. Although irisin is known as an exercise-induced myokine, inconsistent results have been reported in humans (11, 16, 17).
A meta-analysis indicated that chronic exercise training leads to a significant reduction in irisin concentration in the randomized controlled trials, while evidence remains inconclusive in the non-randomized studies (22). Another meta-analysis reported that irisin concentration increased immediately after an acute session of exercise in adults (40). In these two mentioned papers, the irisin level alterations were studied following three (AT + RT + concurrent training) or two types (AT + RT) of exercise.
In the present study, the effect of AT and RT on irisin concentration was examined separately. In line with our results, a study observed that acute or chronic endurance exercise did not affect irisin concentration (41). It has been recognized that AT induced PGC-1α, which has an important role in irisin production. Some evidence has pointed to independent irisin raise and these studies require to be more explored and validated (8).
Moreover, lean mass, fat mass, and glucose concentration have been observed to be associated with irisin levels (42). It has been shown that irisin concentration is positively correlated with an enhanced risk of insulin resistance and metabolic diseases (5). Therefore, it has been reported that irisin is not only released by muscles but also by adipose tissue. It seems that adipose tissue reduction by AT could affect irisin concentration (43). In other words, a reduction in adipose tissue as an irisin target tissue and the origin of irisin secretion can modulate its concentration (7). It is a candidate to treat obesity and type 2 diabetes and can elevate insulin sensitivity by diminishing endoplasmic reticulum stress and improving the survival of pancreatic beta cells and GLUT4 (42).
Irisin promotes fat browning by upregulating the uncoupling protein 1 (UCP1) expression (4). It can stimulate mitochondrial biogenesis by improving the expression of PPARA and mitochondrial transcription factor A (TFAM) (2, 3). It has been reported that irisin can modulate the function of diverse organs, such as cardiac and skeletal muscles, liver, bone, brain, adipose tissue, pancreas, ovary, and kidney at different levels (7).
Irisin not only enhances the oxidative capacity of muscle but also can stimulate muscle hypertrophy by affecting the Akt/mTOR/p70S6 pathway (44). Although it is released from various tissues, the skeletal muscles can secrete more than 70% of irisin in humans (45). Consequently, an increase in muscle mass by RT seems to affect irisin concentration (8). Consistent with our findings, a study observed that eight-week RT can elevate irisin level, while AT did not change its concentration significantly. RT is suggested as an efficient exercise type in overweight/obese people considering the increased irisin concentration (10).
Zhao et al. (2017) found serum irisin increment after 12-weeks of RT (35). Meantime, cross-sectional investigations have determined that irisin concentration is positively associated with insulin-like growth factor-1 (IGF-1), and the strength and circumference of biceps (44, 46, 47). In comparison, the irisin level is negatively correlated with age, cholesterol, insulin, and adiponectin concentration. Irisin potential has been shown in metabolic homeostasis (9, 44). Similarly, irisin raise has been reported through IGF-1 secretion during RT which promotes cell proliferation and myogenesis (44, 46, 48).
With all this in mind, it has been reported that skeletal muscle development by RT can increase myokines release (49, 50). Although the upregulation of PGC1α is an essential factor for irisin release, intracellular muscle ATP deprivation by muscle contraction and SMAD2/3 suppression by follistatin seem to stimulate irisin secretion following RT (2, 3). However, there is no sufficient knowledge to elucidate the mechanism of exercise-induced irisin.
Furthermore, the present study had some limitations. Firstly, several studies have been performed on animals and there is little knowledge about humans. Second, our ability was limited due to the low number of investigations which have studied acute or chronic AT and RT effects on irisin concentration in human. Third, some studies have used several types of medications but our primary search was limited to English ones. Therefore, we were not able to extract all data and to obtain all potentially relevant studies.
Conclusion
The current review and meta-analysis showed that SLTAT, STAT, and LTAT cannot affect irisin concentration. However, it was found that LTRT can augment irisin concentration.
Acknowledgements
This study is based on research project No. 46208, which was sponsored by Ferdowsi University of Mashhad, Iran.
Conflicts of Interest
The authors notified that there are no conflicts of interest about the publication of this article.
Received: 2020/07/14 | Accepted: 2020/09/24 | Published: 2020/12/30
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |